Modern batteries contain highly reactive chemicals that will react at elevated temperature by default. The objective is to operate in a stable environmental bandwidth. Safety is a sensitive issue that gets much media and legal attention, especially with Li-ion batteries.
Any energy storage device carries a risk, and in the 1800s steam engines exploded and people got hurt. Carrying highly flammable gasoline in cars was a hot topic in the early 1900s. Battery makers are obligated to meet safety requirements, but less reputable firms may cheat — it’s “buyers be beware!” Most OEMs use only Li-ion batteries that comply with one or several safety standards.
Li-ion using conventional metal oxides is nearing its theoretical limit on specific energy. Rather than optimizing runtime, battery makers are improving manufacturing methods to enhance safety and increase the calendar life. The real problem lies in rare occasions when an electrical short develops or incorrect charging methods are used.
Let’s examine the inner workings of the cell closer. A mild short will only cause elevated self-discharge and the heat build-up is minimal because the discharging power is very low. If, however, a serious short occurs, a sizable current begins to flow between the electrodes of the cell, and the it heats up.
Uneven separators may also trigger a cell failure. Poor conductivity due to dry areas increases the resistance, which can generate local heat spots that weaken the integrity of the separator. Heat is always an enemy of the battery.
When fully charged, elevated temperature causes a harmful reaction between the positive and negative electrodes and the electrolyte. As a small water leak in a faulty hydro dam can develop to a torrent and take a structure down, so can heat build up damage the insulation layer in a cell and cause an electrical short. The temperature can quickly reach 500°C (932°F), at which point the cell catches fire or explodes. This thermal runaway that occurs is known as “venting with flame.” “Rapid disassembly” is the preferred term by the battery industry.
If the battery gets very hot, immediately remove the device from proximity to flammable materials and bring it to a non-combustible surface. If at all possible, put the device outdoors and let it burn out. If the fire occurs in an airplane the FAA tells flight attendants not to use fire extinguishers but specifies water or pop (soda). Water cools the adjacent material and prevents the fire from spreading. Many research laboratories and factories also use water to put out battery fires. Allow good ventilation while the battery burns itself out. Li-ion contains no lithium metal and does not react with water.
During a thermal runaway, the high heat of the failing cell may propagate to the next cells, causing them to become thermally unstable also. A chain reaction can occur in which each cell disintegrates on its own timetable. A pack can thus be destroyed in a few seconds or over several hours as each cell is being consumed one by one. To increase safety, packs should include dividers to protect the failing cell from spreading to the neighbouring one. (In the Tesla Roadster car, each cell is encased in its own metal compartment.)
Charging and Chargers
The performance and longevity of rechargeable batteries relies on good charging, in today's price conscious market chargers are often given low priority by users, however chargers are ultimately responsible not only for how well your battery performs but how long it lasts and how safe it is too. A good quality charger will ensure that your batteries perform at their best and that they are not damaged or stressed by charging thus increasing their life and decreasing the risk of failure.
A special note on Electronic Cigarette Chargers.
E-cigs use two basic styles of battery, there are the Mechanical Mods and APV's that use standard Li-Ion batteries of the type we have discussed already and there are the integrated all in one "eGo" style batteries that are most often used by beginners. It is these "eGo" style batteries we shall discuss in this section. Ego batteries once disassembled contain a standard Li-Ion battery that is permanently fixed inside the housing. Figure 14 shows the inside of an Ego.

Figure 14: Disassembled eGo Twist.
As with all Li ion batteries care must be taken when charging. It is very important that you use the correct charger for your battery. DO NOT MIX AND MATCH!. using the incorrect charger can result in the battery exploding.
We must also dispel the Myth about Apple iPad / iPod chargers. if you use the correct charger dongle for your battery then it is of little difference if the USB supply that powers the charger has an output rating of 1000mA or 500mA. It is the charger that regulates the batteries charging, not the supply that powers the charger. If in any doubt about the charging dongle you have then replace it with one bought from a reputable dealer (EBay is not a reputable dealer - EBay is simply a market place and like any market there is good and bad available).

In the interest of safety do not use any charger that looks like this - see Figure 15, they are unregulated and when used with Ego style batteries will result in the battery exploding.
Figure 15:
USB Charger socket marketed for E-cigs
Charging Li Ion batteries
Before we discuss charging in detail we should cover the topic of the C Rate
What is the C-rate?
In the late 1700s, Charles-Augustin de Coulomb ruled that a battery that receives a charge current of one ampere (1A) passes one coulomb (1C) of charge every second. In 10 seconds, 10 coulombs pass into the battery, and so on. On discharge, the process reverses. Today, the battery industry uses C-rate to scale the charge and discharge current of a battery.
Most portable batteries are rated at 1C, meaning that a 1,000mAh battery that is discharged at 1C rate should under ideal conditions provide a current of 1,000mA for one hour. The same battery discharging at 0.5C would provide 500mA for two hours, and at 2C, the 1,000mAh battery would deliver 2,000mA for 30 minutes. 1C is also known as a one-hour discharge; a 0.5C is a two-hour, and a 2C is a half-hour discharge.
The battery capacity, or the amount of energy a battery can hold, can be measured with a battery analyzer. The analyzer discharges the battery at a calibrated current while measuring the time it takes to reach the end-of-discharge voltage. An instrument displaying the results in percentage of the nominal rating would show 100 percent if a 1,000mAh test battery could provide 1,000mA for one hour. If the discharge lasts for 30 minutes before reaching the end-of-discharge cut-off voltage, then the battery has a capacity of 50 percent. A new battery is sometimes overrated and can produce more than 100 percent capacity; others are underrated and never reach 100 percent even after priming.
When discharging a battery with a battery analyzer capable of applying different C‑rates, a higher C‑rate will produce a lower capacity reading and vice versa. By discharging the 1,000mAh battery at the faster 2C, or 2,000mA, the battery should ideally deliver the full capacity in 30 minutes. The sum should be the same as with a slower discharge since the identical amount of energy is being dispensed, only over a shorter time. In reality, internal resistance turns some of the energy into heat and lowers the resulting capacity to about 95 percent or less. Discharging the same battery at 0.5C, or 500mA over two hours, will likely increase the capacity to above 100 percent.
Li ion batteries are often given a maximum discharge rating expressed as a C Rate. To convert the batteries C Rate to Amps you need to know it's capacity in mAh (milliamp hours). The rate is the mAh rating / 1000 multiplied by the C rate. so a 2000mAh battery with a 4c rating can supply 2000/1000x4 so 2000 divided by 1000 =2, multiply that by the C rating of 4 and you get a discharge rate of 8Amps.
It should be noted that the C Rate is a constant discharge current meaning that that is the highest current that the battery can supply continuously. There is also something known as a burst current, which is the absolute highest current that the battery can safely supply, usually for only a few seconds and usually the battery will need to rest before it can supply that current again. This is usually only used by people who use these batteries for sub ohm vaping. More information on burst currents can be obtained from the batteries manufacturer and the author of this document does not recommend using batteries in such a manner.
Charging speed and chargers
Charging and discharging batteries is a chemical reaction, but Li-ion is claimed as an exception. Here, battery scientists talk about energies flowing in and out as part of ion movement between anode and cathode. This claim has merits, but if the scientists were totally right then the battery would live forever, and this is wishful thinking. The experts blame capacity fade on ions getting trapped. For simplicity, we consider aging a corrosion that affects all battery systems.
The Li‑ion charger is a voltage-limiting device that is similar to the lead acid system. The difference lies in a higher voltage per cell, tighter voltage tolerance and the absence of trickle or float charge at full charge. While lead acid offers some flexibility in terms of voltage cut‑off, manufacturers of Li‑ion cells are very strict on the correct setting because Li-ion cannot accept overcharge. The so-called miracle charger that promises to prolong battery life and methods that pump extra capacity into the cell do not exist here. Li-ion is a “clean” system and only takes what it can absorb. Anything extra causes stress.
Most cells charge to 4.20V/cell with a tolerance of +/–50mV/cell. Higher voltages could increase the capacity, but the resulting cell oxidation would reduce service life. More important is the safety concern if charging beyond 4.20V/cell. Figure 16 shows the voltage and current signature as lithium-ion passes through the stages for constant current and topping charge.
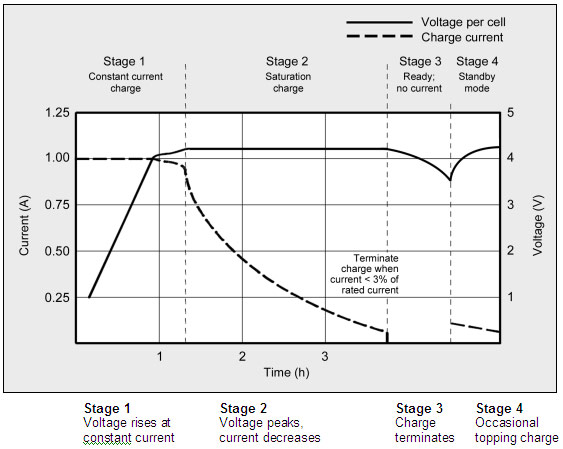
Figure 1: Charge stages of lithium-ion. Li-ion is fully charged when the current drops to a predetermined level or levels out at the end of Stage 2. In lieu of trickle charge, some chargers apply a topping charge when the voltage drops to 4.05V/cell (Stage 4).
Courtesy of Cadex
The charge rate of a typical consumer Li-ion battery is between 0.5 and 1C in Stage 1, and the charge time is about three hours. Manufacturers recommend charging the 18650 cell at 0.8C or less. Charge efficiency is 97 to 99 percent and the cell remains cool during charge. Some Li-ion packs may experience a temperature rise of about 5ºC (9ºF) when reaching full charge. This could be due to the protection circuit and/or elevated internal resistance. Full charge occurs when the battery reaches the voltage threshold and the current drops to around three percent of the rated current. A battery is also considered fully charged if the current levels off and cannot go down further. Elevated self-discharge might be the cause of this condition. As can be seen from the graph the voltage only rises during the early part of the charge cycle and should never exceed 4.2v.
Increasing the charge current does not hasten the full-charge state by much. Although the battery reaches the voltage peak quicker with a fast charge, the saturation charge will take longer accordingly. The amount of charge current applied simply alters the time required for each stage; Stage 1 will be shorter but the saturation Stage 2 will take longer. A high current charge will, however, quickly fill the battery to about 70 percent.
Li-ion does not need to be fully charged, as is the case with lead acid, nor is it desirable to do so. In fact, it is better not to fully charge, because high voltages stresses the battery. Choosing a lower voltage threshold, or eliminating the saturation charge altogether, prolongs battery life but this reduces the runtime. Since the consumer market promotes maximum runtime, these chargers go for maximum capacity rather than extended service life.
Some lower-cost consumer chargers may use the simplified “charge-and-run” method that charges a lithium-ion battery in one hour or less without going to the Stage 2 saturation charge. “Ready” appears when the battery reaches the voltage threshold at Stage 1. Since the state-of-charge (SoC) at this point is only about 85 percent, the user may complain of short runtime, not knowing that the charger is to blame. Many warranty batteries are being replaced for this reason, and this phenomenon is especially common in the cellular industry.
Avoiding full charge has benefits, and some manufacturers set the charge threshold lower on purpose to prolong battery life. Table 2 illustrates the estimated capacities when charged to different voltage thresholds with and without saturation charge
Overcharging Lithium-ion
Lithium-ion operates safely within the designated operating voltages; however, the battery becomes unstable if inadvertently charged to a higher than specified voltage. Prolonged charging above 4.30V forms plating of metallic lithium on the anode, while the cathode material becomes an oxidizing agent, loses stability and produces carbon dioxide (CO2). The cell pressure rises, and if charging is allowed to continue the current interrupt device (CID) responsible for cell safety disconnects the current at 1,380kPa (200psi).
Should the pressure rise further, a safety membrane bursts open at 3,450kPa (500psi) and the cell might eventually vent with flame. The thermal runaway moves lower when the battery is fully charged; for Li-cobalt this threshold is between 130–150C°C (266–302°F), nickel-manganese-cobalt (NMC) is 170–180°C (338–356°F), and manganese is 250°C (482°F). Li-phosphate enjoys similar and better temperature stabilities than manganese.
Lithium-ion is not the only battery that is a safety hazard if overcharged. Lead- and nickel-based batteries are also known to melt down and cause fire if improperly handled. Nickel-based batteries have also been recalled for safety concerns. Properly designed charging equipment is paramount for all battery systems.
What sort of charger should I use?
Only use a charger specifically designed for use with Li Ion batteries, Purchase your charger from a reputable dealer and keep it well maintained. There are currently 3 well recognised and trusted brands of charger. They are Xtar, Nitecore, and Efest. All three manufacturers make a range of models ranging from simple single cell chargers to ones capable of holding 6 cells at a time. If you use one of these chargers ensure you read the user manual thoroughly and that you maintain your charger correctly.
Some devices may use a specific charger suitable only for that device (e.g.: eGo batteries), do not use the charger for anything else and only charge the device with the recommended charger.
Some devices may be equipped with a USB port for charging and this can be used with any USB supply, some of these devices allow use as a "pass-through" allowing the device to be powered by the USB port whilst charging. At first glance this seems an excellent idea but in practice using the pass-through option results in significantly longer charge times.
General battery care
Batteries are energy stores, to release that energy safely they need to be looked after.
Always keep Li Ion batteries away from metal objects during storage. if storing your batteries for long periods bring them to about half charge, it will reduce stress on the battery and help them last longer.
Li Ion batteries should never be discharged below 3.2v or charged above 4.2v.
Check your batteries for damage before use, if the case is deformed, or damaged or the wrapper is damaged do not use it.
Always check that the battery terminals and contacts are clean to get the best connection.
If your battery gets unusually hot during charging stop charging immediately and take the battery outdoors if possible until it cools, then dispose of it safely.
Keep batteries in a plastic case designed for the purpose when not in use.
Never leave batteries unattended when charging. You may want to invest in a safe charging bag.
Toby Kilroy
Writer at POTVToby has been vaping since early 2012 and has used an array of devices and kit in that time. He sometimes writes up reviews but is often found with his head stuck in pages of code with a confused smile on his face. Toby also helps run his wife's site gethistory.co.uk and has two children. He sometimes fondly remembers having free time and occasionally manages to sneak away to put his head into a good book!
Join the discussion
Electricity & Ohm's Law
A basic introduction in order to develop and appreciation for electricity and to build safe coils.
Battery Configurations
To stack or not to stack. Tubbyengineer answers your question...